Preparation of 1-Bromobutane
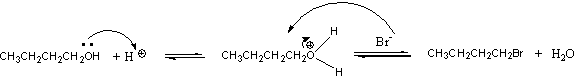
The mechanism is SN2.
CAUTION: Wear gloves during the entire
experiment
Bring your 100 mL RBF, wide stem funnel, and cork ring to one of the balances.
Add 17 gm. NaBr to the flask, then proceed over to the tray with the water and
1-butanol repipettors.
Add 15 mL H2O and 10 mL 1-butanol to the flask and return to your hood workstation.
At your hood workstation, clamp the flask to the ringstand attached to your
hotplate/stirrer, add your magnetic stirring bar, and begin stirring the contents of the flask.
Bring a 50 or 100 mL beaker to the front of the lab and dispense 15 mL conc. H2SO4
Return to your hood workstation and S L O W L Y AND CAREFULLY the conc. H2SO4
to the flask.
Equip the flask with a reflux condenser and begin circulating water through it - water going in the
bottom and out the top of the condenser.
(
see the figure on the next page )
Heat the flask gently (using the hot plate at a setting of about “4” ).
Continue gentle heating until the mixture begins to reflux. (liquid dripping from the condenser )
Once this is observed, continue "refluxing" the mixture
for 60 minutes, controlling the
level of the vapor in the condenser so
that it reaches no more than half-way up the
condenser
|
|
At the end of the 60 minute reflux period, discontinue heating
Add 700 mL of water to your 1L beaker
Once the contents of the flask ceases to bubble, remove the reflux condenser, unclamp the flask
from the ringstand and pour the contents of the flask into the Separatory funnel.
Immediately separate the lower aqueous layer from the organic layer and add it to the contents of your 1 L Beaker, WITH STIRRING. (Why is the aqueous layer on the bottom ? )
Extract
the organic layer with 15 mL H2O.
Separate the
UPPER aqueous layer from the
organic layer and add it to the contents
of your 1 L Beaker, WITH STIRRING.
Extract the organic layer with 15mL saturated sodium bicarbonate.
Drain the lower ORGANIC layer into a DRY 50 mL Erlenmeyer flask, and
add CaCl2 .
Swirl the flask occasionally for a period of 5 minutes. ( Why was the organic layer now on the bottom ? )
Allow the drying agent to settle and DECANT the liquid into a TARED sample bottle,
appropriately
labeled. Save it in your locker for
the alkyl halide tests.
Notes:
Steps 7-13: Initially, the reaction is exothermic which makes the reaction occur, but
quickly loses the energy, so the energy must be supplied by the heating
mantle in order to complete the reaction as much as possible.
Refluxing is a means of trapping the vapors of the reactants and cooling
them enough to return to the reaction flask. There, they have another
opportunity to react to form 1-bromobutane
Steps 17-20: To separate the 1-bromobutane, a series of extractions are performed. To
determine which layer is the 1-bromobutane layer, you take a test tube and add
5 mL of water to it, then remove a few drops of the bottom layer from the
separatory funnel and see if they fall through the water to the bottom. If they do,
the bottom layer is the 1-bromobutane layer or organic layer, if not, the upper
layer is the organic layer. To confirm your decision, remove a few drops of the
top layer in the separatory funnel with a pasteur pipette and place them in a dry
test tube and add 2 to 3 mL of water, dropwise, to see if it falls to the bottom.
The larger layer should be the water layer.
Step 25: Washing with water simply removes any unreacted H2SO4 molecules.
Step 26: Washing with saturated NAHCO3 removes any remaining H2SO4.molecules.
Step 28: The water is removed by CaCl2, as described in the propanoic acid
experiment.
